
Phosphorus Lewis Dot Structure Drawing, Several Compounds And Detailed
A single phosphate is linked to two organic groups is called phosphate diester. The backbone of DNA is linked by phosphate diesters. Organic phosphates are often abbreviated using ' OP O P ' and ' OPP O P P ' for mono- and diphosphates, respectively. For example, glucose-6-phosphate and isopentenyl diphosphate are often depicted as shown below.

PO4 3 Lewis Structure The Phosphate Ion YouTube
Example 8.3. 1: Benzene. Benzene is a common organic solvent that was previously used in gasoline; it is no longer used for this purpose, however, because it is now known to be a carcinogen. The benzene molecule ( C 6 H 6) consists of a regular hexagon of carbon atoms, each of which is also bonded to a hydrogen atom.
:max_bytes(150000):strip_icc()/phosphate-anion-58c046973df78c353ca2b689.jpg)
List of Common Polyatomic Ions
3PO 4 Phosphoric acid [H 2PO 4]− Dihydrogen phosphate [HPO 4]2− Hydrogen phosphate [PO 4]3− Phosphate or orthophosphate In organic chemistry, phosphate or orthophosphate is an organophosphate, an ester of orthophosphoric acid of the form PO 4RR′R″ where one or more hydrogen atoms are replaced by organic groups.
CH105 Chapter 5 Introduction to Organic Chemistry Chemistry
This chemistry video tutorial explains how to draw the lewis structure of PO4 3-, the phosphate ion. It also discusses the formal charge and resonance struc.
PO4 3 Lewis Structure How to Draw the Lewis Structure for PO43 YouTube
How to Draw Lewis Structure of PO4 3- I Easy & Quick Science Genius 26 subscribers Subscribe 0 2 views 9 months ago This video will explain how to draw a skeletal structure of phosphate.

Simple Method for writing Lewis Structures of the phosphate ion(PO4)3
When drawing Lewis dot structures, the overall charge on a polyatomic ion is equal to the sum of the formal charges on each atom in the ion.. phosphate is a polyatomic ion that always has a charge of 3-. Since the overall charge for an ionic compound is zero, we can use the chemical formula and the charge on phosphate to calculate the charge.
Identify The Lewis Structure Resonance Forms For Po That My XXX Hot Girl
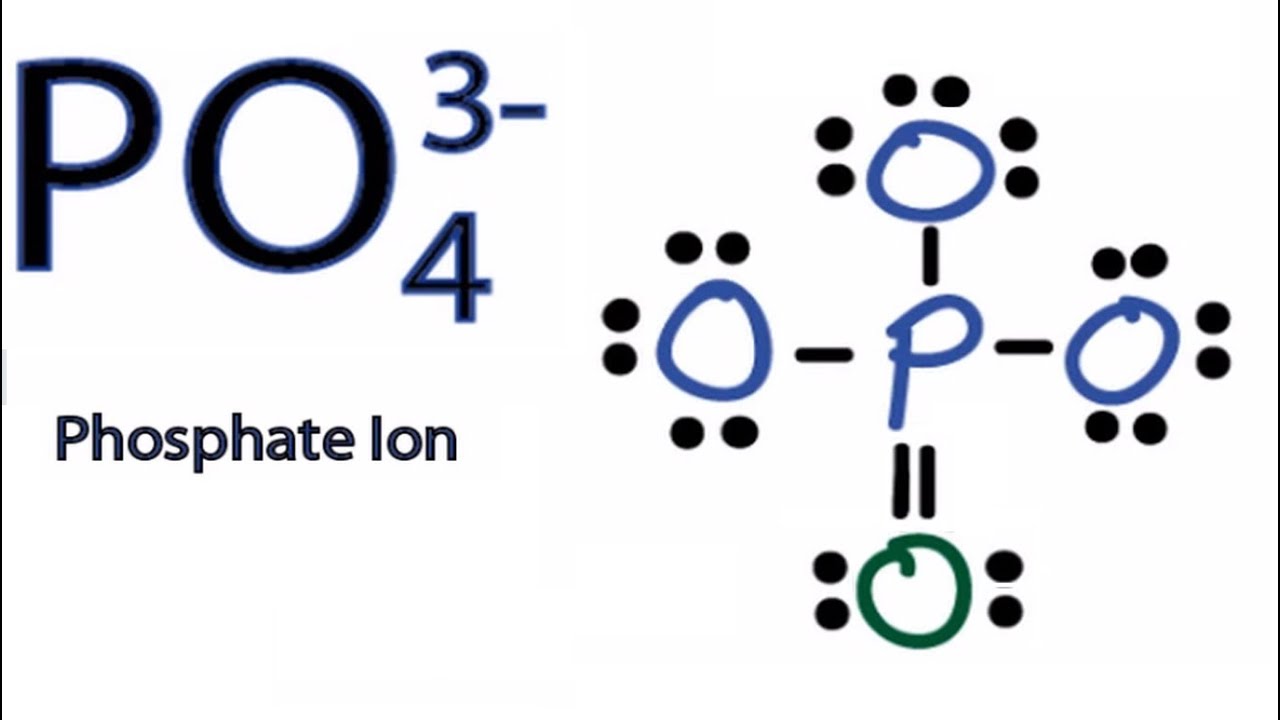
Drawing the Lewis structure of the phosphate ion involves representing the central phosphorus atom surrounded by four oxygen atoms. Each oxygen atom is connected to the phosphorus atom by a single bond, and the remaining electrons are placed as lone pairs.

Phosphate Anion Chemical Structure Photograph by Molekuul/science Photo
A step-by-step explanation of how to draw the PO43- Lewis Dot Structure (Phosphate ion).For the PO4 3- structure use the periodic table to find the total num.

Phosphate
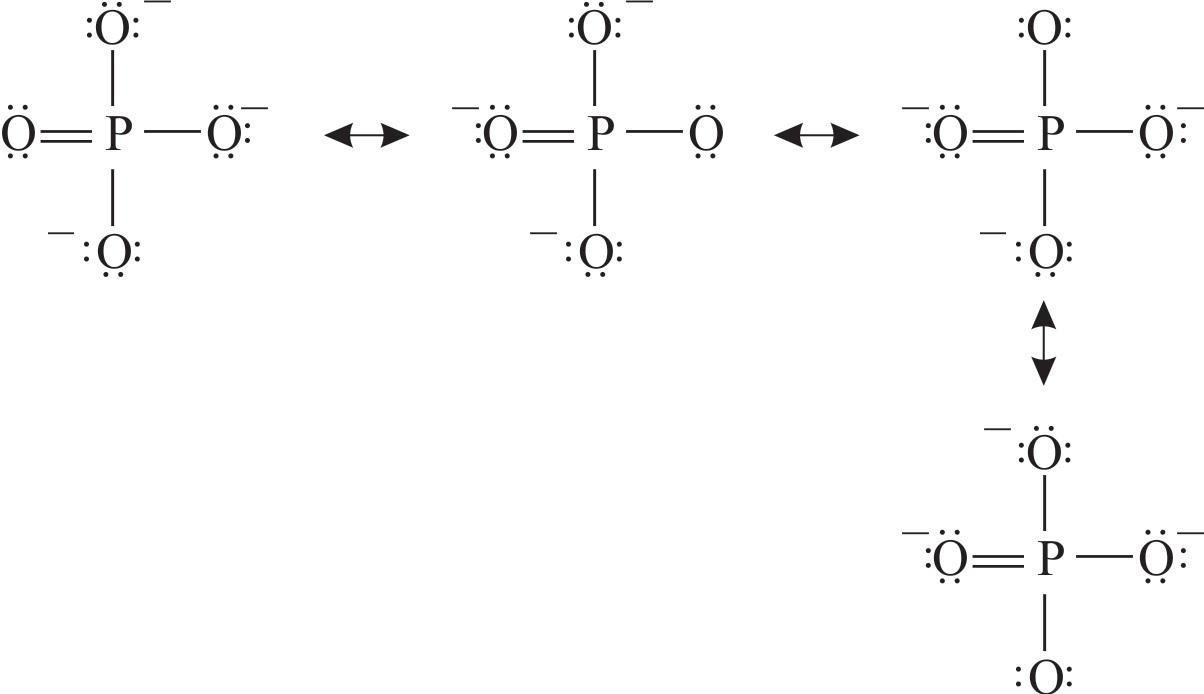
The structure on the right is the Lewis electron structure, or Lewis structure, for H 2 O. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet.. Resonance structures are particularly common in oxoanions of the p-block elements, such as sulfate and phosphate, and in aromatic hydrocarbons, such as benzene and.

Phosphorus Definition, Facts, Symbol, Discovery, Property, Uses
Steps of drawing PO4 3- lewis structure Step 1: Find the total valence electrons in PO4 3- ion. In order to find the total valence electrons in PO4 3- ion (phosphate ion), first of all you should know the valence electrons present in phosphorus atom as well as oxygen atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)
41. What is the Lewis dot structure of phosphate ion? how many
This widget gets the Lewis structure of chemical compounds. Send feedback | Visit Wolfram|Alpha Get the free "Lewis Structure Finder" widget for your website, blog, Wordpress, Blogger, or iGoogle. Find more Chemistry widgets in Wolfram|Alpha.
Electron Shell 015 Phosphorus Phosphorus Electron Dot Diagram , Free
1 Answer Sorted by: 6 Calcium phosphate crystallises in a number of different modifications of which β- CaX3(POX4)X2 C a X 3 ( P O X 4) X 2 is the stable room temperature and ambient pressure modification. It has a rather complex structure not easily described, consisting of chains and rings with phosphate and calcium ions in specific patterns.

Draw the Lewis Dot structure for the Phosphate anion?… SolvedLib
To draw the Lewis structure of any compound, we always use the concept of valence electrons (number of electrons in the outer shell of each atom). The overall charge on the phosphate ion is -3 and the phosphorus atom is in an oxidation state of +5.

Lewis Dot Structure of Phosphate (PO4 3).....No More Confusion
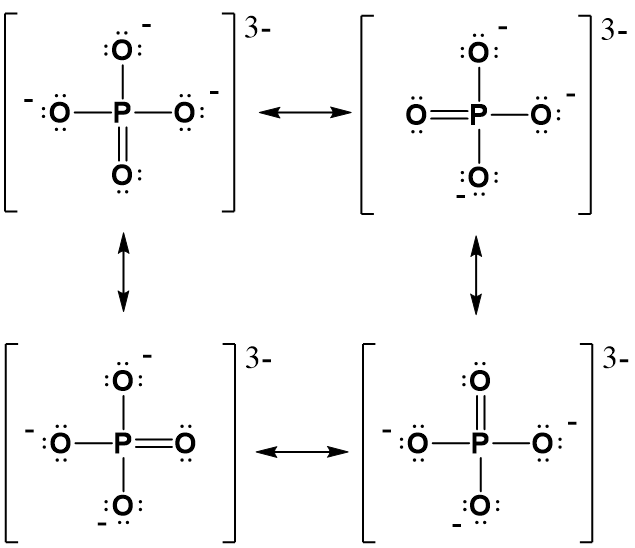
In lewis structure, there should be charges on atoms. Phosphate ion | PO 43- Phosphate ion is one of the oxyanion of phosphorous. Phosphorous is at +5 oxidation state in PO 43-. Also, phosphate ion has a -3 charge. Lewis structure of PO 43- ion In the lewis structure of PO 43-, three is a double bond between phosphorous atom and one oxygen atom.

Phosphorus Lewis Dot Structure Drawing, Several Compounds And Detailed
1.2.2 Lewis Structures of Polyatomic Molecules or Ions.. Figure 1.2m phosphate anion Lewis structure. Elements in Period 3 (or higher) have 3 (or more than 3) principle shells, so the d orbital is available in the valence shell. That is why they can accommodate more than 8 electrons.
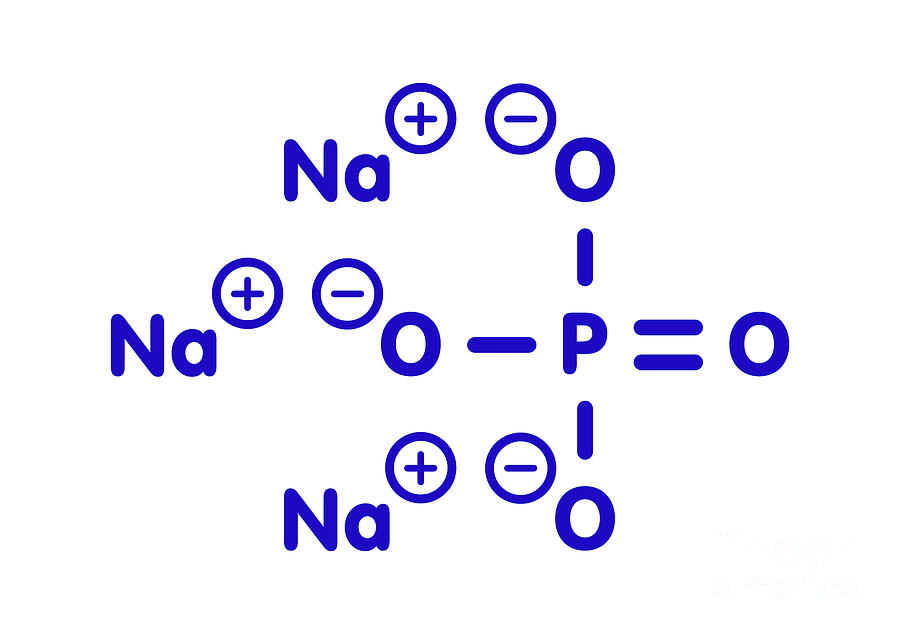
Sodium Phosphate Chemical Structure Photograph by Molekuul/science
Share 994 views 10 years ago This is a tutorial on how to draw a Lewis dot structure (diagram) of a phosphate ion.more.more This is a tutorial on how to draw a Lewis dot structure.