Diagrama De Bohr Tabla
Bohr model of all Elements is mentioned in the chart below.. Phosphorus (P) 2, 8, 5: 16: Sulfur (S) 2, 8, 6: 17: Chlorine (Cl) 2, 8, 7: 18: Argon (Ar) 2, 8, 8: 19: Potassium (K) 2, 8, 8, 1: 20:. Orbital Diagram of All Elements (Diagrams given Inside) Subscribe to our newsletter. Subscription Form.
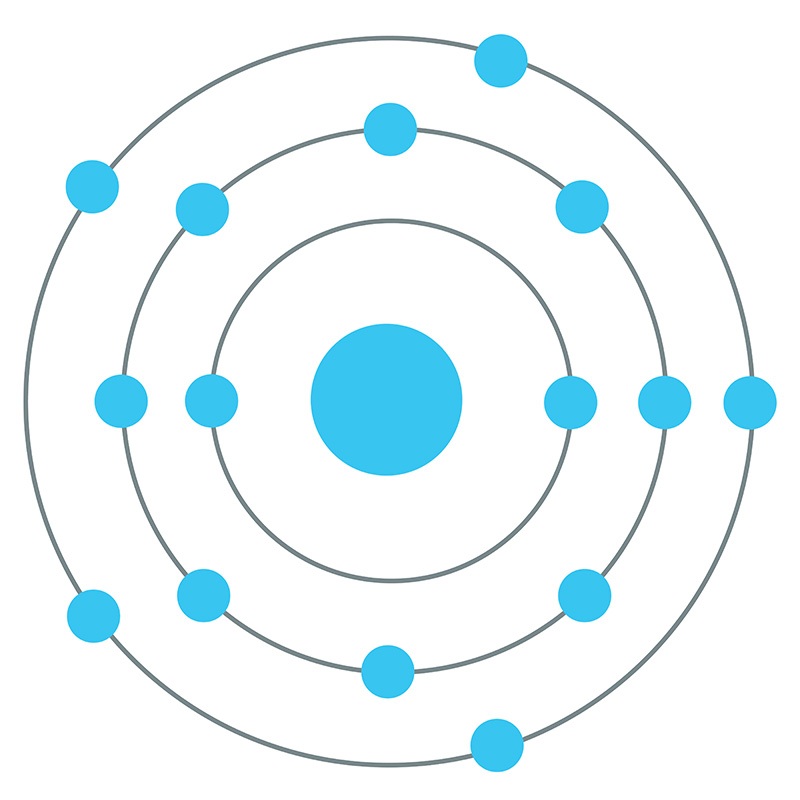
Phosphorus Bohr Diagram
Argon has 2 electrons in its first shell, 8 in its second, 8 in its third.Check me out: http://www.chemistnate.com

Bohr Model Phosphorus Atom Electron Structure Stock Vector (Royalty
We explain how the Bohr atomic model works and provide example Bohr diagrams. Call Direct: 1 (866) 811-5546 Sign In Start Free Trial. the Rutherford Model. The Bohr Model has an atom with a positively-charged nucleus surrounded by negatively-charged electrons that have circular, planetary-like orbits.. Phosphorus (P) 15: 15: 16: Sulfur (S.
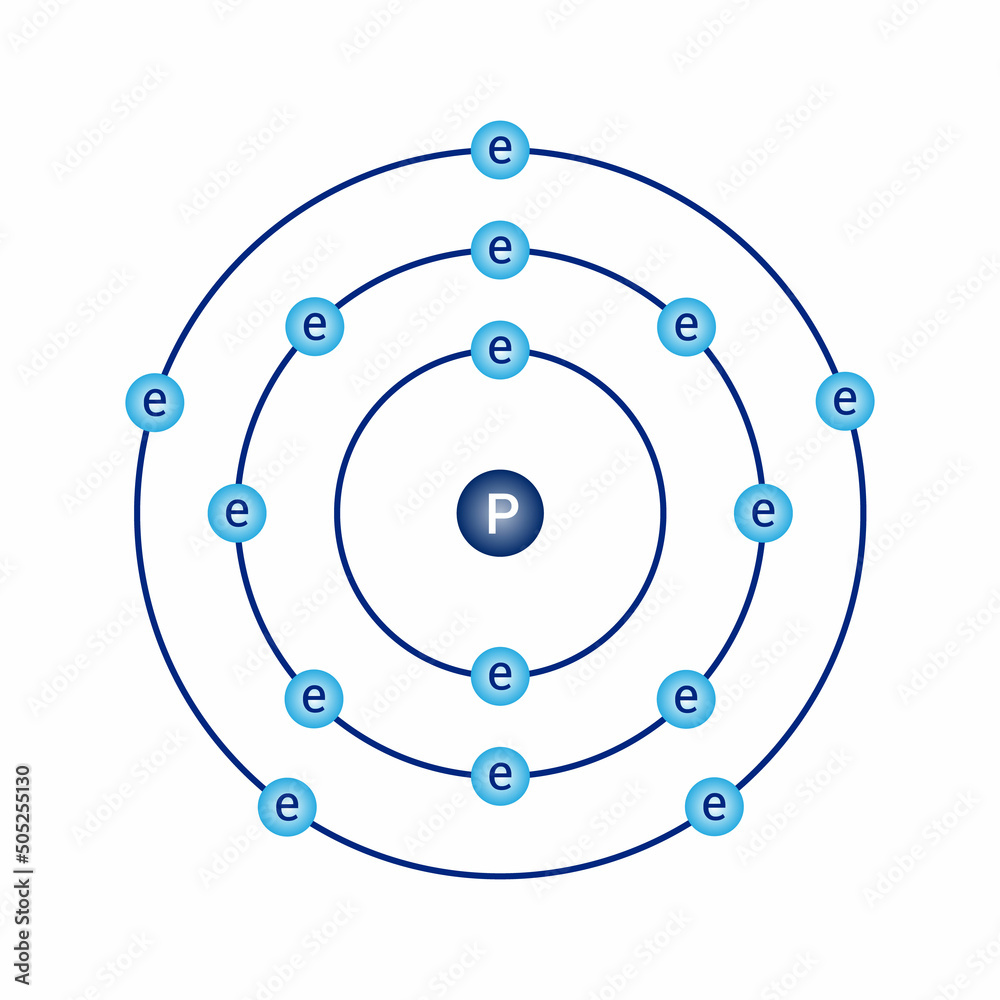
Bohr model diagram of phosphorus P in atomic physics vector de Stock
Numerous models of the atom had been postulated based on experimental results including the discovery of the electron by J. J. Thomson and the discovery of the nucleus by Ernest Rutherford. Bohr supported the planetary model, in which electrons revolved around a positively charged nucleus like the rings around Saturn—or alternatively, the.

Phosphorus Bohr Model — Diagram, Steps To Draw Techiescientist
4) The following diagram is a Bohr-Rutherford diagram of one element from the periodic table: To which group and period does this element belong? A) Period 3 group 4. B) Period 4 group 4. C) Period 3 group 1. D) Period 1 group 3. 5) Referring to the periodic table, find an element that has the same number of electron shells and
draw your own bohr model for dickson only Tutor help Now
The electrons occur in three orbits. Two electrons are in Orbit 1, eight electrons are in Orbit 2 and five electrons are in Orbit 3. The outer orbit (i.e., valence) is not full. Thus, phosphorus will react with other elements to reach the maximum. Example 2: Fluorine This is the Bohr -Rutherford Diagram for Fluorine (Atomic Number 9).

Pin on phosphorus
Find step-by-step Biology solutions and your answer to the following textbook question: (a) Draw the Bohr-Rutherford diagram (without neutrons) for an atom of each of the following elements: lithium, oxygen, calcium; and phosphorus. (b) Draw the; Bohr-Rutherford diagram (without neutrons) for the ion formed by each of the elements in (a). (c) Write the chemical symbol for each ion.
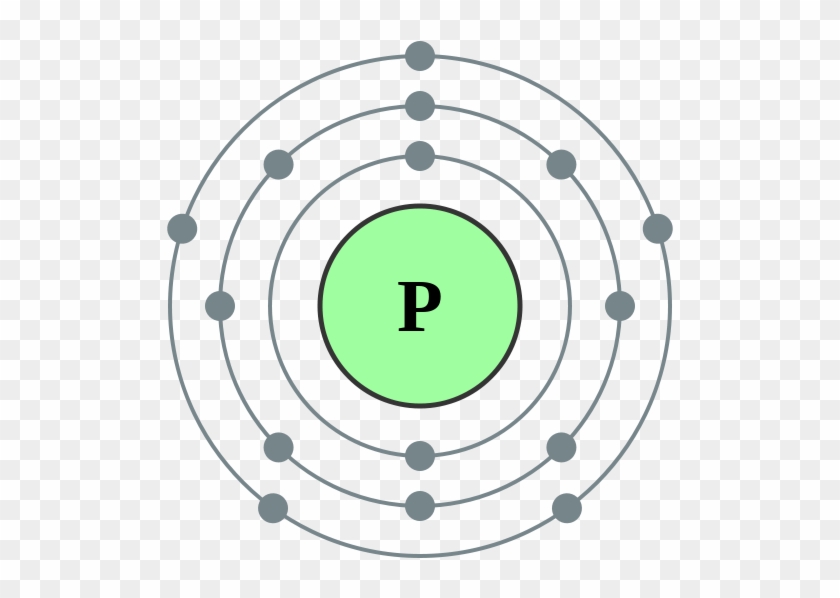
Bohr Model Of A Phosphorus Atom Electron Configuration Of Sodium
Niel Bohr, in 1913, presented the Rutherford model with the required corrections, and this model since forth has been recognized as the Bohr-Rutherford model of the atom.. Below is the video attached explaining how to draw a Bohr model diagram of a Phosphorus atom. Conclusion.
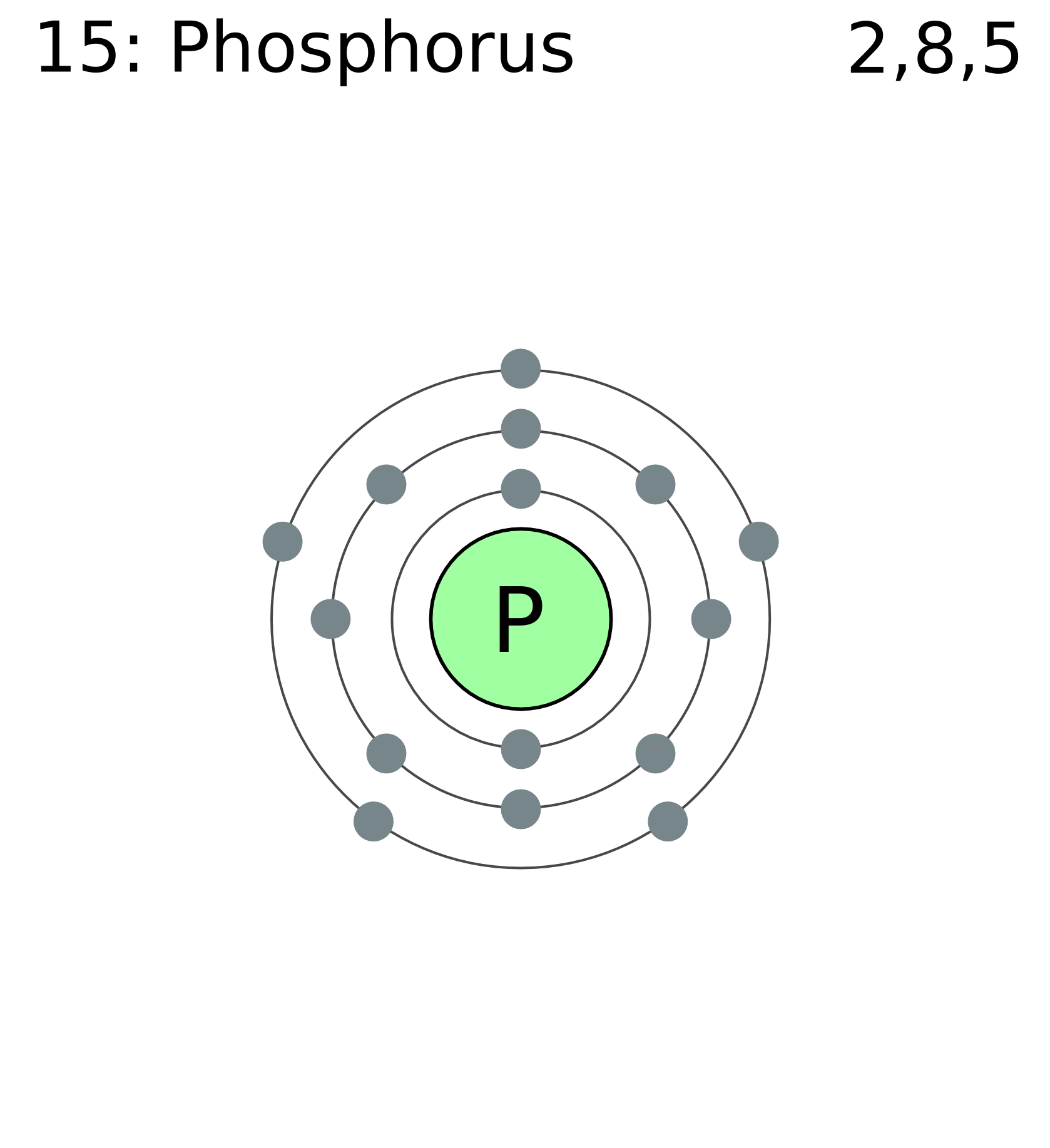
Phosphorus Electron Configuration (P) with Orbital Diagram
How to draw the Bohr-Rutherford Diagram for Phosphorous. 2 electrons can go in the first shell, 8 in the second, 8 in the third, and so on.
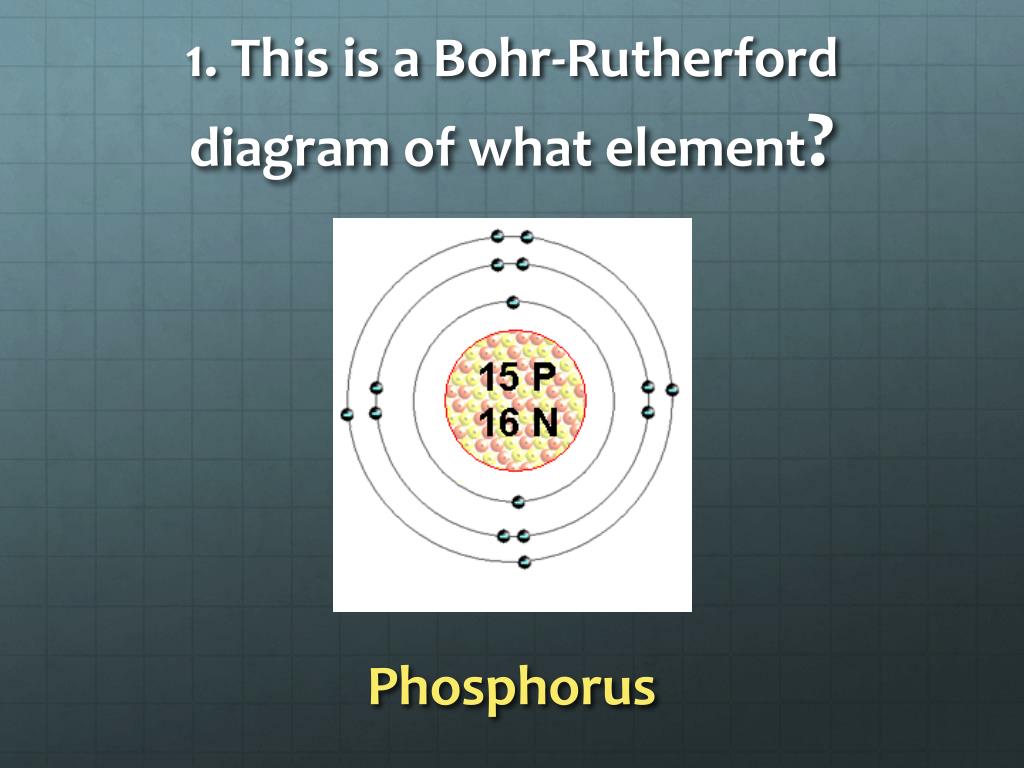
PPT SNC2D CHEM UNIT REVIEW PowerPoint Presentation, free download
Figure \(\PageIndex{1}\): A Bhor's model can be used to diagram the location of electrons in each energy shell for an atom. Notice that protons go in the nucleus of the atom and electrons are drawn on orbits surrounding the nucleus. Image from Wikimedia commons. Example \(\PageIndex{2}\) Draw the Bohr's model for sodium (Na).
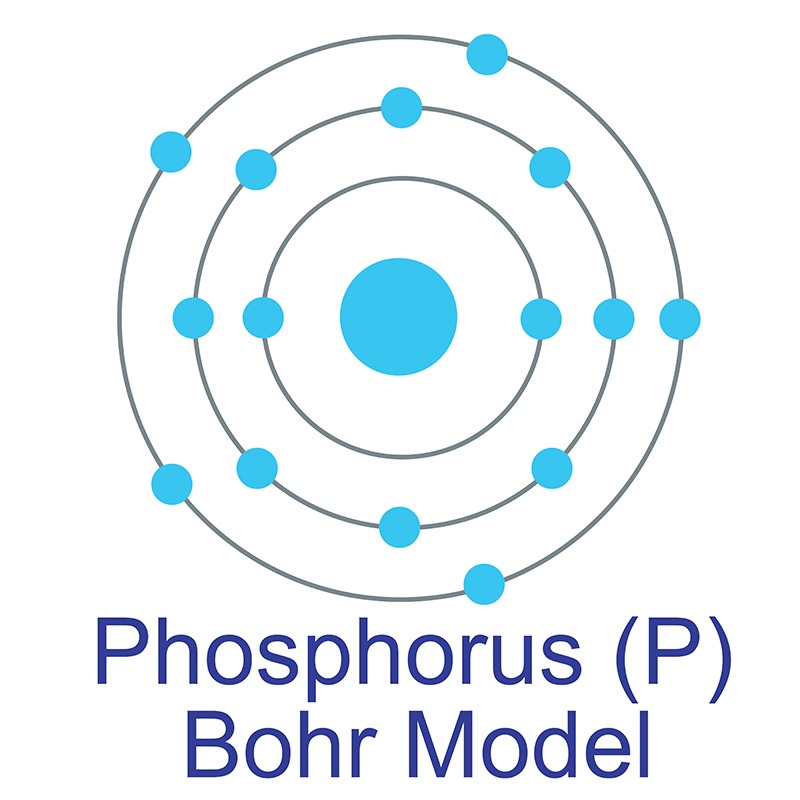
Phosphorus Bohr Diagram
Find step-by-step Biology solutions and your answer to the following textbook question: How are the Bohr-Rutherford diagrams of nitrogen and phosphorus similar and how are they different?.
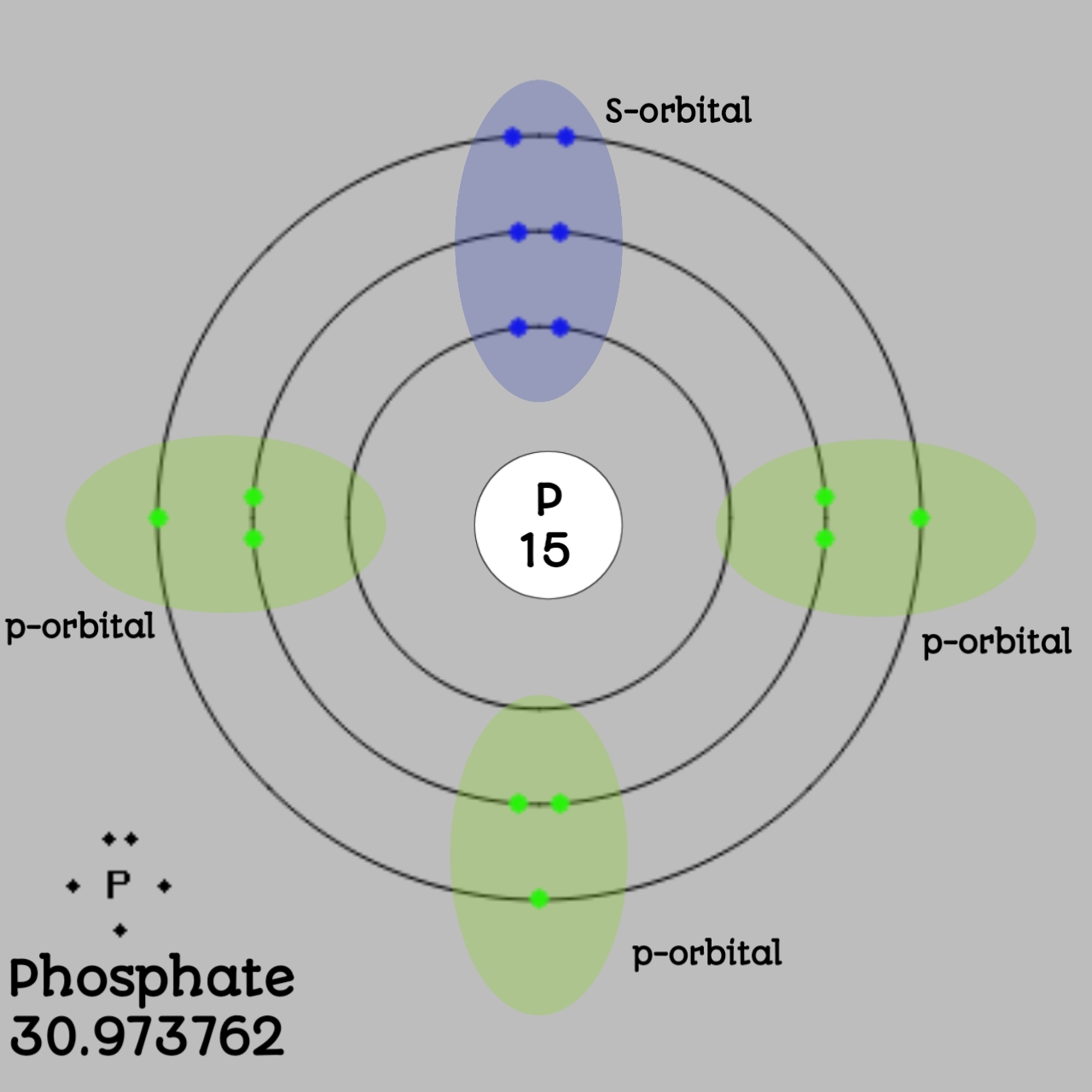
Phosphorus Bohr Diagram
(a) Draw the Bohr-Rutherford diagram (without neutrons) for an atom of each of the following elements: lithium, oxygen, calcium; and phosphorus. (b) Draw the; Bohr-Rutherford diagram (without neutrons) for the ion formed by each of the elements in (a). (c) Write the chemical symbol for each ion.

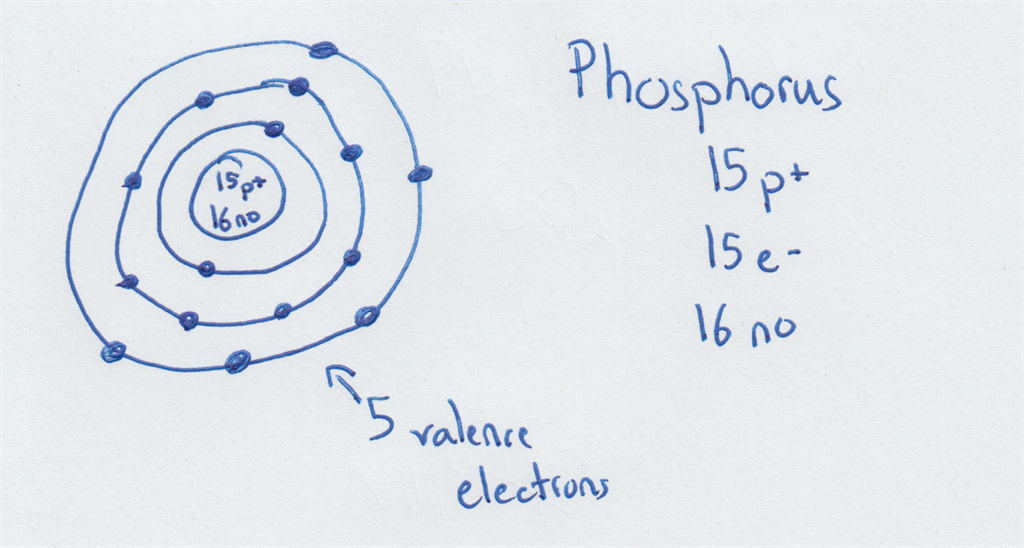
Number Of Valence Electrons In Phosphorus
Drawing Bohr-Rutherford diagrams is super easy using the following steps: Find the number of protons, neutrons and electrons for the atom. The number of protons is the atomic number. The number of neutrons can be found by subtracting the number of protons from the atomic mass rounded to the nearest whole. This is because protons and neutrons.
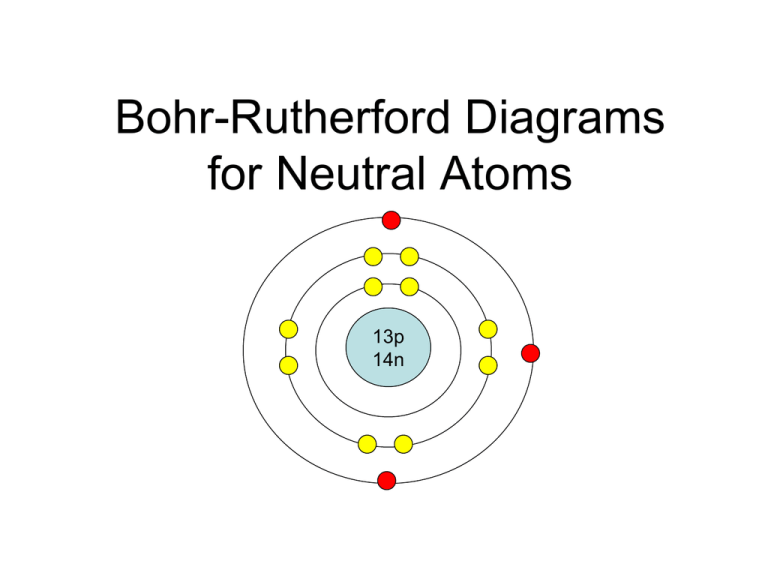
BohrRutherford diagrams for atoms
The Bohr - Rutherford Diagram also shows the total number of electrons is 15. Recall: The number of protons = number of electrons. The electrons occur in three orbits. Two electrons are in Orbit 1, eight electrons are in Orbit 2 and five electrons are in Orbit 3. The outer orbit (i.e., valence) is not full. Thus, phosphorus will react

How many valence electrons are in an atom of Phosphorus MakeTheBrainHappy
- The Bohr-Rutherford model of the atom **Please note that the entire unit including 12 power points and 238 slides, can be purchased from my store for the price of 5 single lessons**. Periodic Table and Bohr-Rutherford Diagram Lesson Lewis Structures Ions Ionic Compounds Polyatomic Ions and Compounds Molecular Compounds Chemical Reactions
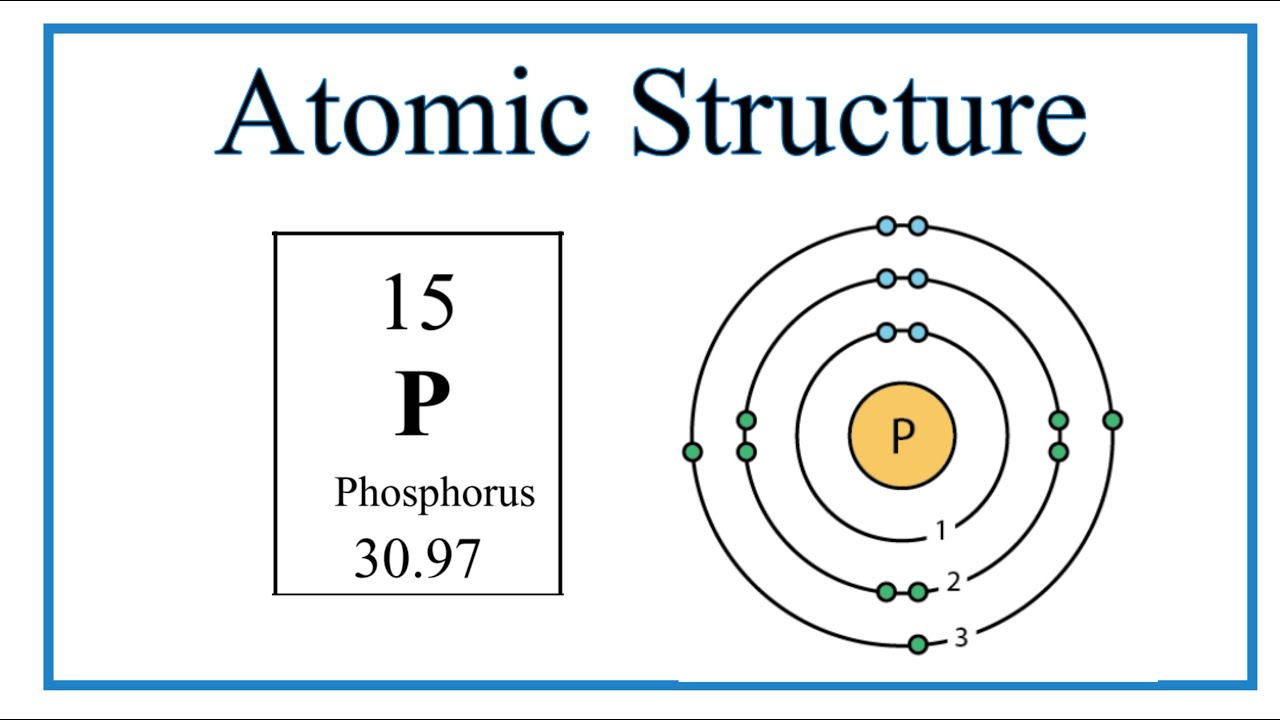
Atomic Structure (Bohr Model) for Phosphorus (P) YouTube
The Bohr Model of Phosphorus(P) has a nucleus that contains 16 neutrons and 15 protons. This nucleus is surrounded by three-electron shells named K-shell, L-shell, and M-shell. The outermost shell in the Bohr diagram of Phosphorus contains 5 electrons that also called valence electrons.